What Is a Graphite Solution and Why Modern Industries Need It?
Nov 20, 2025
Introduction
The term graphite solution has become common across industries that depend on high-performance carbon and graphite materials. Companies like SGL, Mersen, Toyo Tanso, and many global graphite specialists describe their services not as "graphite products," but as graphite solutions. This shift reflects a deeper trend: industrial customers no longer buy simple blocks or components. They buy results, performance, stability, and engineering support.
As a company with more than 25 years of experience in specialty graphite and carbon materials, SHJ CARBON works with clients from semiconductor, high-temperature metallurgy, chemicals, glass, photovoltaic processing, battery manufacturing, and more. From our global experience, one insight remains constant:
Before understanding a graphite solution, you must first understand graphite itself-its structure, its properties, its variations, and its industrial roles.
Only then can engineers, buyers, and manufacturers understand why the term "solution" matters so much.
What Does "Graphite Solution" Mean?

A graphite solution is not simply a material. It combines:
- material selection
- grade recommendation
- engineering design
- precision machining
- purification
- coating (SiC, PyC, etc.)
- performance matching
- long-term application support
This explains why major carbon companies use the term. Industrial environments differ widely in temperature, atmosphere, load, purity requirements, and corrosion exposure. A single graphite grade rarely fits all conditions. A graphite solution provider helps customers select the right graphite, not the most expensive one.
At SHJ CARBON, we define a graphite solution as:
The process of matching the right graphite material, processing method, and coating to the customer's real application, based on engineering judgment and long-term experience. This approach reduces cost, extends component life, and ensures consistent performance.
What Is Graphite?
To understand graphite solutions, you first need a clear and accurate picture of what graphite really is. Graphite is an allotropic form of carbon in which each carbon atom bonds to three neighboring carbon atoms in a flat, sp²-hybridized hexagonal network. The fourth electron remains delocalized above and below each layer, which gives graphite its high electrical and thermal conductivity.
These hexagonal carbon sheets stack on top of each other and form layers. Inside each layer, the C–C bonds are strong and stiff; between layers, only weak van der Waals forces hold them together. This contrast creates graphite's typical behavior:
- Very strong and stiff in the plane of the layers
- Easy to shear and lubricious between layers
Most industrial graphite is not a single crystal but a polycrystalline material. It consists of many small graphite crystallites, pores, and binder phases. As a result, the "same" graphite grade can show very different performance if you change:
- the raw material (petroleum coke, pitch coke, natural graphite)
- the forming process (isostatic pressing, molding, vibration forming, extrusion)
- the graphitization temperature and time
- any impregnation, purification, or coating treatment
Because of these factors, two graphite blocks that look similar can have very different density, porosity, strength, electrical resistivity, and service life-and therefore a very different price. This is exactly why industrial users do not just need graphite; they need a graphite solution that matches the right material structure to the real working conditions.
Types of Graphite Used in Industrial Applications
For engineers working in high-temperature testing or industrial heat treatment, electric resistance isn't just a secondary spec-it's one of the core parameters that defines thermal field performance.
Natural Graphite
Natural graphite forms over millions of years inside the earth's crust. It starts as carbon-rich organic material-such as plant matter or sediment-that gets buried and subjected to:
- high temperature
- high pressure
- long-term geological stress
Under these conditions, the carbon atoms slowly rearrange into the layered hexagonal structure that we call graphite. Differences in:
- temperature profile
- pressure level
- surrounding minerals
- fluid movement

lead to different natural graphite types:
- Flake graphite – plate-like crystals in metamorphic rocks
- Vein (lump) graphite – high-purity graphite in veins and fissures
- Amorphous graphite – fine, microcrystalline material mixed with other minerals
Because natural graphite comes from geological processes, its:
- purity (ash content)
- crystal size
- density and porosity
- structural uniformity
can vary a lot from deposit to deposit-even within the same mine. This variability shapes its application window. Natural graphite works well where: bulk performance matters more than tight tolerance. some variation in structure is acceptable
Typical uses include:
- refractory bricks and castables for iron and steel
- foundry facings and coatings
- brake linings and friction materials
- lubricants and greases (especially flake graphite)
- expandable graphite for flame-retardant systems
certain battery anodes where cost is a key factor and structure can be managed by additional processing However, for high-precision graphite components-for example, semiconductor fixtures, vacuum furnace hot zone parts, or complex machined blocks-natural graphite usually cannot offer:
- the required dimensional stability
- the necessary purity level
- the controlled porosity and grain size
This is why most engineered graphite solutions for critical applications rely on artificial (synthetic) graphite instead of natural graphite.
Artificial Graphite
To understand why the industry often talks about graphite solutions, you first need to understand how artificial graphite is made. Unlike natural graphite-which forms over millions of years deep underground-artificial graphite is an engineered material created through a precise, multi-step industrial process.
Every performance characteristic-density, strength, electrical resistivity, porosity, thermal stability-comes from how it is manufactured.
This section explains the logic behind each stage so engineers and buyers can understand why different graphite grades exist and why their properties vary so widely.
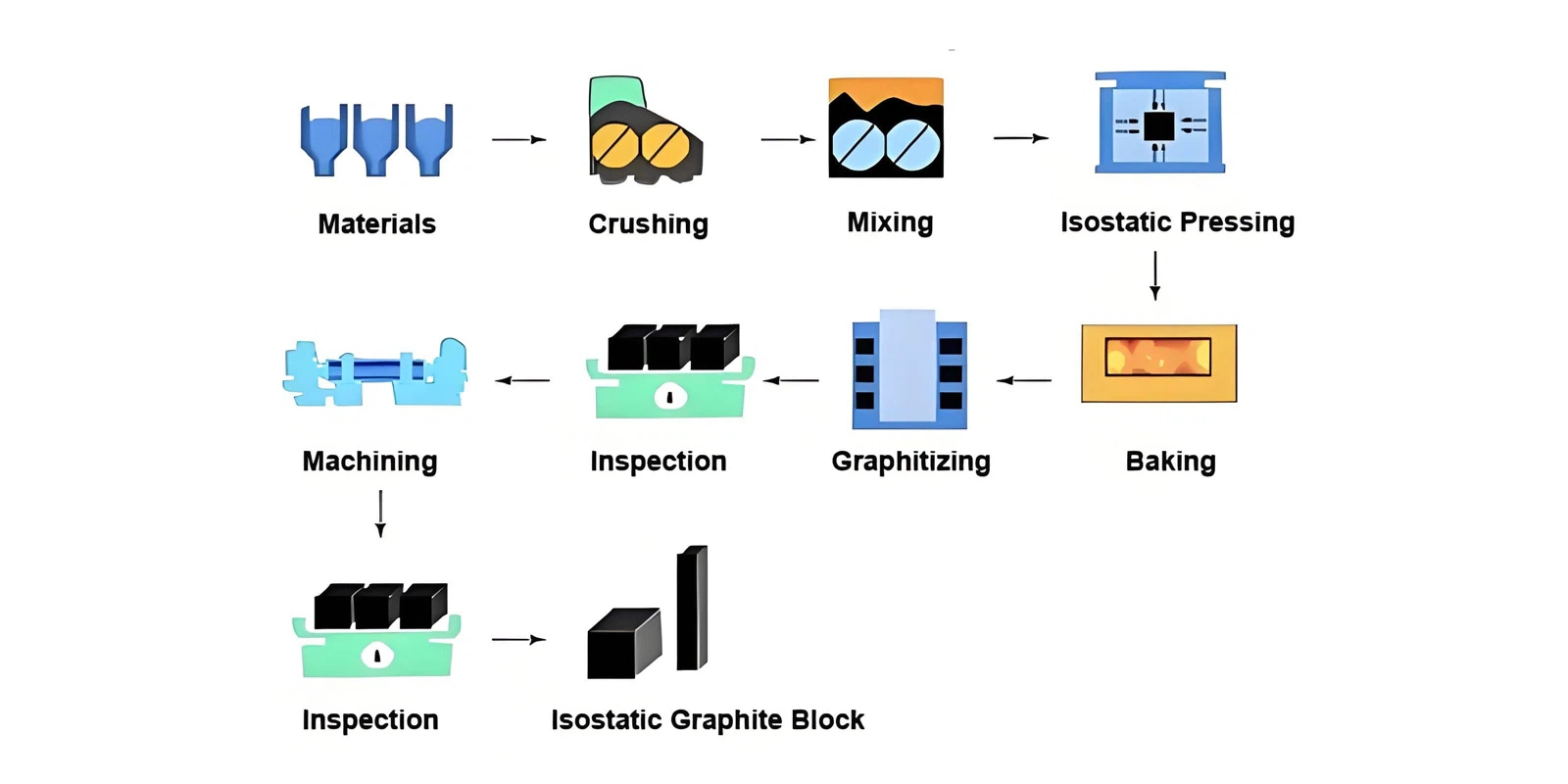
1. Raw Materials: Where Artificial Graphite Begins
Artificial graphite uses carbon-rich raw materials such as:
- petroleum coke
- needle coke (for high-end grades)
- pitch coke
These raw materials serve as the aggregate, the solid particles that form the structure of the final graphite. Their particle size, purity, and microstructure directly influence the characteristics of the final product. For example:
- Large particle sizes → lower density, more anisotropy
- Ultra-fine particles → high density, ideal for isostatic graphite
Raw materials also include a binder, typically coal tar pitch, which softens and coats the aggregates so they can be shaped.
2. Crushing and Particle Classification
Raw coke must be crushed into specific particle-size distributions. This step is fundamental because particle size affects:
- packing behavior
- porosity
- binder absorption
- strength
Different forming methods require different particle sizes:
- Extruded graphite → larger particle size
- Molded graphite → fine to medium particles
- Isostatic graphite → ultra-fine particles (often < 0.3 mm)
A precise particle-size recipe ensures consistent structure in the final material.
3. Mixing: Creating a Uniform Carbon Mixture
After crushing, aggregates are mixed with binder in a heated mixer. The binder melts and coats every particle, forming a uniform mixture known as green paste.The ratio of aggregate to binder depends on:
- target density
- forming method
- strength requirements
Additional additives may be included:
- graphite scrap → improves thermal behavior
- natural graphite → improves lubrication
- carbon black → improves conductivity
This stage establishes the fundamental microstructure.
4. Forming: The Step That Defines Material Directionality
The forming method determines whether graphite will be anisotropic or isotropic. Each forming technique produces a distinct internal structure, which determines how the final material behaves under heat, pressure, or mechanical load.
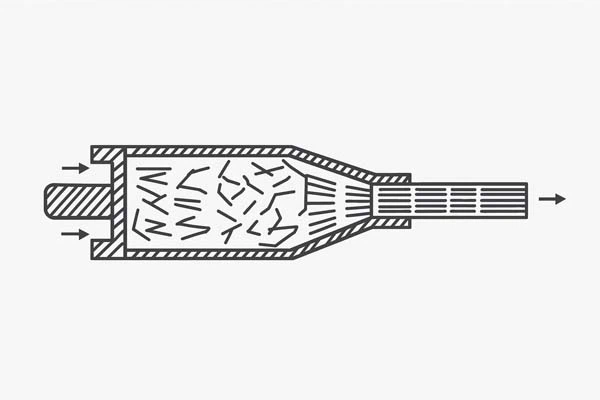
Extrusion (Extruded Graphite)
- Paste is forced through a die
- Particles align in the extrusion direction
- Material becomes anisotropic
- Suitable for rods, tubes, long products

Molding (Die-Pressing)
- Powder is pressed inside a rigid mold
- Directionality is weaker but still present
- Suitable for blocks and small precision parts

Isostatic Pressing (CIP)
- Pressure applies from all directions simultaneously
- Particle packing becomes uniform
- Produces isotropic graphite
- Used for semiconductor, EDM, high-temp furnace parts
5. First Baking: Turning Binder into Carbon
The shaped "green body" is baked slowly at 700–1200°C, sometimes for several weeks. During baking:
- binder carbonizes
- volatile components evaporate
- the block shrinks
- pores form
This converts the mixture into a solid carbon body, but not yet graphite. The slow heating rate is crucial, especially between 400–600°C, where internal stresses can cause cracks if not controlled.
6. Impregnation: Increasing Density and Strength
After baking, the carbon body contains pores. For applications requiring:
- high density
- low permeability
- better mechanical strength
- improved oxidation resistance
the block is placed into a high-pressure vessel (autoclave) and impregnated with:
- pitch
- resin
- or other carbonizable materials
Some grades undergo multiple impregnation–rebaking cycles until the required density is reached.
7. Second Baking: Carbonizing the Impregnated Material
A second baking step carbonizes the impregnated materials, further increasing density and structural stability.
This second baking is faster than the first since only the impregnated binder needs carbonizing.
At this stage, the material becomes dense carbon, ready for the next crucial step.
8. Graphitization: Transforming Carbon Into Graphite
Graphitization is the defining step of artificial graphite production. The carbon block is heated to 2800–3000°C in a graphitization furnace. At this temperature:
- carbon atoms realign into hexagonal graphite layers
- electrical resistivity decreases
- thermal conductivity increases
- the material becomes machinable
- dimensional stability improves drastically
Different manufacturers apply different temperatures, heating rates, and cycle durations-leading to differences in quality and cost. Graphitization is the main reason synthetic graphite can outperform natural graphite in high-precision or high-temperature environments.
9. Purification and Special Treatments
Depending on the application, the graphite may undergo additional treatments:
High-temperature halogen purification
Removes impurities down to 1–5 ppm for:
- semiconductor equipment
- nuclear graphite
- high-vacuum furnace components
- Resin or metal impregnation
Improves properties such as:
- oxidation resistance
- gas tightness
- friction characteristics
- machinability
These treatments tailor the final properties to specific industrial needs.
Why Understanding This Process Matters
Artificial graphite is not a single material-it is a family of engineered materials. Two blocks may look identical but perform completely differently because:
- raw materials differ
- particle sizes differ
- forming methods differ
- baking and graphitization temperature differ
- impurity levels differ
This is why the industry emphasizes graphite solutions rather than generic "graphite products." Graphite is engineered for purpose, not chosen at random.
Understanding the Reason Behind Multiple Graphite Grades

Industrial buyers often wonder: "Why does graphite come in so many grades, codes, and price levels?" The answer lies in its structure and processing. Graphite's properties change dramatically based on:
- raw materials (pitch coke vs petroleum coke)
- forming method (isostatic > molded > vibration molded > extruded)
- graphitization temperature
- impregnation cycles
- purity level
- grain size
- porosity
- electrical resistance
- thermal conductivity
Two blocks of graphite may look identical, yet one might cost three times the other because it performs far better in high-temperature or corrosive environments.
As SHJ CARBON's senior materials engineer Frank often says: "A material is never simply 'good' or 'bad.' It is only suitable or unsuitable for a given application."This is the essence of a graphite solution.
Key Properties That Make Graphite a Solution-Oriented Material
Properties of graphite
In addition to the regular products that we already produce.
Light Weight with High Strength
Despite its solid appearance, graphite remains light. Its density ranges from 1.55–1.95 g/cm³, making it ideal for high-temperature and structural applications where weight matters.
Extremely High Melting Point (~3500°C)
Graphite withstands temperatures that most metals cannot. This is why graphite plays essential roles in:
- foundry operations
- high-temp furnaces
- SiC crystal growth
- chemical vapor deposition
Its stability at extreme temperatures makes it irreplaceable.
Excellent Electrical & Thermal Conductivity
Graphite conducts both heat and electricity extremely well. This enables its use in:
- electrodes
- battery anodes
- heaters
- thermal distribution components
- electronic contacts
Graphite's conductivity originates from its mobile electrons between layers.
Natural Lubrication
The layered structure slides smoothly, creating outstanding self-lubrication. This reduces friction in:
- mechanical systems
- wheels
- industrial seals
- high-temperature contact surfaces
Chemical Stability & Corrosion Resistance
Graphite withstands:
- acids
- alkalis
- corrosive gases
- reactive metals
This makes it ideal for chemical reactors, heat exchangers, and containers handling aggressive environments.
Anisotropic Mechanical Behavior
Graphite behaves differently depending on direction:
- strong in-plane
- weaker between layers
This directional behavior enables engineered performance in precision devices like EDM electrodes, sintering molds, or semiconductor fixtures.
Where Graphite Is Used in Modern Industry

Graphite particles help eliminate friction and protect surfaces.


Graphite withstands molten steel, iron, and glass, making it essential in foundries.

Used in motor brushes, electrodes, and grounding systems.

High-purity graphites and SiC-coated graphite play critical roles here.

Graphite acts as a neutron moderator due to its atomic structure.

High-purity graphite serves as the source material.



Why Buyers Often Feel Confused About Graphite
Many customers say:
"Why does every supplier give me different grade names?"
"Why is the price difference so large?"
"Why do American codes, German codes, and Chinese codes look unrelated?"
This confusion arises because:
- Different countries use different graphite naming conventions
- Graphite isn't standardized like steel
- Performance depends on manufacturing process, not name
- Suppliers often promote their own proprietary grades
Graphite must be evaluated by engineering indicators, not just names. This is why buyers need a graphite solution, not a catalog.
Why Graphite Solutions Exist

Industries don't need materials; they need performance. A graphite solution provider helps customers:
- choose the right materials
- analyze application needs
- balance cost vs performance
- design components
- perform precision machining
- apply purification or coating
- verify usage through testing
- close the loop with data and feedback
A true graphite solution requires expertise, experience, and engineering judgment.
How SHJ CARBON Provides Graphite Solutions
SHJ CARBON has been in the graphite and carbon materials field for more than 25 years. Our team includes engineers with decades of experience in specialty graphite, purification, coating, and application engineering. We support customers in the full value chain:
- Material Selection:Matching graphite grades to actual application conditions.
- Precision Machining:Complex 3D components with tight tolerances.
- Purification:Up to 5–10 ppm purity levels for semiconductor applications.
- Coating:SiC, PyC, and other functional coatings extend component life.
- Application Engineering:Understanding heat flow, temperature zones, corrosive gases, or mechanical loads.
- Testing and Feedback:Ensuring real-world performance aligns with engineering expectations.
- Cost Optimization:Recommending alternatives when high-end materials are unnecessary.
We believe the value of a graphite solution lies not in the price of the graphite itself, but in how well it fits the customer's problem.
Case Example: Semiconductor & SiC Industry

Semiconductor processing requires:
- ultra-high temperature
- ultra-low contamination
- tight dimensional stability
- corrosion resistance
Our expertise helps customers balance purity, coating thickness, thermal uniformity, and cost.
Graphite solutions here include:
- graphite susceptors
- wafer carriers
- heater elements
- insulation parts
- SiC-coated graphite components

Conclusion: A Graphite Solution Is Engineering, Not a Product
Graphite's unique structure and broad industrial relevance make it one of the most valuable materials in modern manufacturing. But its complexity also makes it difficult for buyers to choose correctly. A graphite solution:
- clarifies material confusion
- reduces unnecessary cost
- improves product life
- strengthens process stability
- gives customers predictable performance
This is why industries seek graphite solution providers, and why SHJ CARBON continues to support global customers with engineering-driven graphite expertise.







