What Is the Thermal Conductivity of Graphite and Why It Varies?
Dec 23, 2025
Introduction
When people look up the thermal conductivity of graphite, they may be trying to do different things: get a reliable number for reference, compare graphite with metals like copper, or understand why graphite can behave like a strong heat spreader in one direction yet a thermal barrier in another. That mix of questions is exactly what makes graphite interesting-and also easy to misunderstand if we treat thermal conductivity as a single "fixed value."
In SHJ CARBON's day-to-day material discussions, the most useful starting point is not only "How many W/m·K?" but also "In which direction does heat need to move, and under what temperature and atmosphere?" Graphite's thermal performance is closely tied to its microstructure and anisotropy-the same underlying structure logic discussed in our earlier note on isotropic vs anisotropic behavior-so the same material family can show very different results depending on grade and use conditions.
In this article, we'll explain graphite thermal conductivity in a way that works for both quick learning and practical selection: what values to expect, why direction matters, how temperature and structure influence heat transfer, and what this means for real applications.
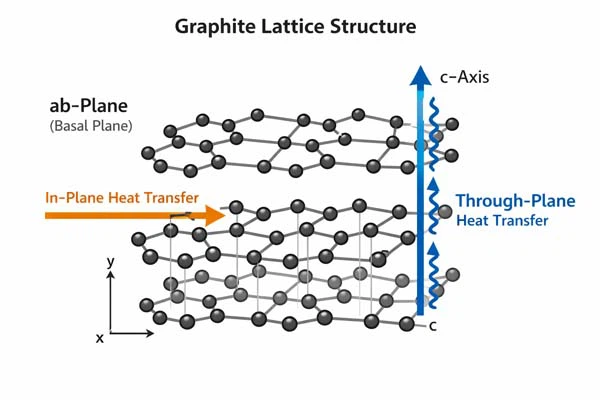
Graphite Thermal Conductivity by Crystal Direction
Graphite exhibits strong anisotropy in thermal conductivity due to its layered crystal structure. Heat transfer primarily occurs through lattice vibrations, or phonons, within the crystal lattice.
ab-plane vs c-axis
The thermal conductivity of graphite differs drastically between the in-plane (ab) and out-of-plane (c-axis) directions:
| Material Type | ab-plane (W/mK) | c-axis (W/mK) |
|---|---|---|
| High-crystallinity pyrolytic graphite | 390–4180 | 2 |
| Commercial pyrolytic graphite | 200–400 | 2 |
| Asphalt-based graphite fiber | 1180 | N/A |
| Copper | 385 | N/A |
| Silver | 420 | N/A |
| Diamond (Type II) | 2000–2100 | N/A |
Thermal conductivity in ab vs c direction
(schematic of lattice vibration amplitude).
In the ab-plane, phonons can travel with minimal scattering, resulting in high thermal conductivity. In contrast, along the c-axis, phonon transport is restricted, reducing thermal conductivity by approximately 200 times.
Impact of Crystallinity and Defects
The thermal conductivity depends strongly on crystal quality. High-crystallinity pyrolytic graphite exhibits near-ideal phonon transport, whereas commercial graphite contains grain boundaries and defects that scatter phonons, reducing thermal conductivity.
Key formula (Debye model):
K=b⋅Cp⋅v⋅L
Where:
- K = thermal conductivity
- b = constant
- Cp = specific heat per unit volume
- v = phonon velocity
- L = mean free path of phonons
As temperature rises, atomic vibrations increase, reducing the mean free path LLL and thus slightly decreasing thermal conductivity.
Thermal Properties of Graphite
Specific Heat & Thermal Expansion
Graphite has a moderate specific heat and a low thermal expansion coefficient, making it suitable for high-temperature applications.
| Property | Value (typical) |
|---|---|
| Specific heat (Cp, J/kg·K) | 710–820 |
| Thermal expansion coefficient (α, 10^-6/K) | 4–8 (ab-plane), 25–30 (c-axis) |
| Maximum service temperature | 3000 K |
This combination of high thermal conductivity in-plane and low expansion reduces thermal stress in devices operating at high temperatures.
Thermal Shock Resistance
Graphite's thermal shock resistance is excellent due to its low thermal expansion along the ab-plane. It withstands rapid heating and cooling cycles better than many metals and ceramics, making it ideal for aerospace components, furnace linings, and high-power electronics.
Why Graphite Conducts Heat So Well
The superior thermal conductivity in graphite arises from phonon transport along the basal plane.
- Lattice vibration (phonons): Heat is carried mainly by vibrations of carbon atoms in the hexagonal lattice.
- Phonon scattering: Grain boundaries and defects reduce conductivity, explaining the difference between ideal and commercial graphite.
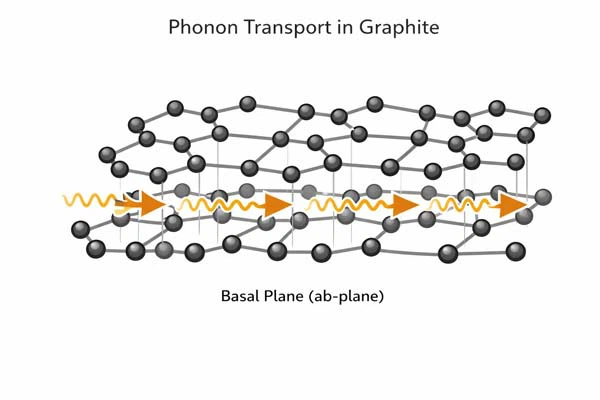
Figure 2: Phonon transport schematic in graphite lattice.
In essence, graphite behaves like a high-performance thermal conductor along the ab-plane, while acting as a thermal insulator along the c-axis, a property exploited in thermal management designs.
Graphite vs Other Materials
Graphite compares favorably with metals and ceramics in thermal conductivity:
| Material | Thermal Conductivity (W/mK) |
| Graphite (ab-plane) | 390–4180 |
| Graphite fiber | 1180 |
| Copper | 385 |
| Silver | 420 |
| Aluminum Nitride | 200 |
| Aluminum Oxide | 25 |
| Diamond (Type II) | 2000–2100 |
Graphite fibers derived from asphalt-based precursors can reach almost three times the thermal conductivity of copper, providing excellent options for lightweight, high-performance heat spreaders.
Applications Leveraging Graphite's Thermal Performance
Graphite's value in thermal design is not just "high conductivity"-it is the ability to engineer heat flow through directional conduction, low mass, and stability under thermal cycling. In many systems, graphite is used either as a heat spreader (moving heat laterally) or as a thermal barrier (reducing heat transfer through thickness), depending on how the microstructure is oriented and how the part is integrated.
Electronics & Heat Management
In electronics, graphite is commonly selected when designers need rapid in-plane heat spreading to reduce hot spots while keeping the assembly lightweight and dimensionally stable.
- Heat spreaders for power devices and modules
Graphite can distribute localized heat away from MOSFET/IGBT/SiC packages to a larger area, helping downstream heatsinks work more efficiently. In practice, performance depends heavily on contact quality (surface flatness, pressure, interface materials) because contact thermal resistance can dominate the thermal path if not managed.
- Thermal interface stacks (TIM + graphite layer)
In real assemblies, graphite rarely works alone. It is often paired with TIMs to bridge micro-gaps and improve heat transfer into a heat spreader. A common design approach is: TIM for contact + graphite for lateral spreading, especially where heat sources are unevenly distributed.
- EV battery thermal management
In battery packs, graphite can help smooth temperature gradients between cells and reduce peak temperatures during fast charge/discharge. The key is clarifying the goal-spreading heat along the plane vs blocking heat through thickness-and selecting the graphite structure accordingly to avoid "good data, weak system effect."
- High-power LEDs and semiconductor cooling
For compact lighting and semiconductor assemblies, hot spots drive color shift and lifetime loss. Graphite heat spreaders are often applied to stabilize junction temperature, but the design must consider heat-flow direction and mounting interfaces, otherwise theoretical conductivity does not translate into real thermal improvement.
Aerospace & Energy Industries
In high-temperature and harsh-duty systems, graphite is valued for its thermal stability, resistance to thermal shock, and predictable behavior under repeated thermal cycling.
- High-temperature insulation & thermal protection
Certain graphite structures are used to control heat leakage in furnaces and thermal protection systems. Here, the priority may be low through-thickness conductivity combined with stability, rather than maximum heat transfer.
- Heat exchangers and structural components in high-temperature zones
Graphite can be used where materials must tolerate heat while maintaining geometry. Selection typically involves balancing thermal conductivity, mechanical strength, and oxidation risk (especially in air at elevated temperatures).
- Energy systems requiring dimensional stability under thermal load
In applications where thermal gradients are unavoidable, graphite's low expansion behavior (in specific orientations/grades) can reduce thermal stress and help maintain alignment. Engineers often evaluate not only conductivity, but also CTE, thermal shock resistance, and machining tolerances.
FAQ – Thermal Conductivity of Graphite
Q1: What is the thermal conductivity of graphite?
A: Varies with type and crystallinity. High-quality pyrolytic graphite can reach 4180 W/mK in the ab-plane, while the c-axis is around 2 W/mK.
Q2: How does graphite compare with copper?
A: In-plane thermal conductivity of graphite can exceed copper, while along the c-axis, graphite is a thermal insulator.
Q3: Why does graphite have high thermal conductivity?
A: Strong covalent bonding and phonon transport in the basal plane enable efficient heat conduction.
Q4: Is graphite a good thermal insulator?
A: Along the c-axis, yes. It can act as a thermal barrier, while in-plane it is a very good conductor.
Q5: How does temperature affect graphite's thermal conductivity?
A: Thermal conductivity decreases slightly with increasing temperature due to phonon scattering.
Conclusion
In practice, thermal conductivity data becomes truly useful only when it helps you make a decision-which graphite grade to choose, how to orient it, and what trade-offs to expect. Whether you are doing a quick comparison for learning or evaluating materials for a real component, the most important step is to connect the numbers to your design goals: heat spreading vs heat blocking, stability over thermal cycles, and performance you can maintain over time.
If you're working through options, a simple way to move forward is to list three items on one line: your application, your temperature range, and how heat needs to travel in the part. Even a short summary like that usually clarifies which parameters matter most and which ones are "nice to have."
If you'd like a second set of eyes, feel free to share those basics-we're happy to point you to the most relevant graphite properties and common selection pitfalls, so you can narrow down choices faster with fewer iterations.







